Abstract
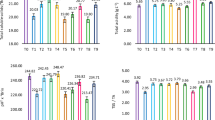
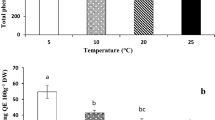
Metabolic changes can occur in plants in response to abiotic stress. Extensive use of leaf discs (mechanical wounding) in studies regarding the effect on the biochemical response of the grapevine to different types of biotic stress makes it necessary to understand metabolic perturbation after injury. In this study, we investigate how mechanical wounding can affect the metabolism of grapevine leaf tissue using Bianca variety as case study. Two sizes of leaf discs (1.1 and 2.8 cm in diameter) were excised from leaves, and phenol, lipid and carotenoid perturbation were investigated 0, 6, 12, 24, 48, 96 and 120 h post cutting. In our study, we found an accumulation of molecules belonging to stilbenoid and stilbene classes such as trans-resveratrol, trans-piceide, Z-miyabenol C, E-cis-miyabenol C and ampelopsin D + quadrangularin A after abiotic stress. The increase in fatty acids such as linoleic acid, linolenic acid and oleic + cis-vaccenic acid during the first 12 h after injury, followed by a return to basal level, allowed us to surmise their role in response to abiotic stress, in particular to mechanical wounding in grapevine leaves. Different-sized discs caused a different response to the tissue, with a higher accumulation in 1.1-cm-diameter discs, especially of phenol compounds. The results of this work can be used to better understand metabolic changes due to biotic stress, having previous knowledge about the perturbation caused by abiotic stress.







Similar content being viewed by others
References
Adrian M, Jeandet P, Veneau J et al (1997) Biological activity of resveratrol, a stilbenic compound from grapevines, against Botrytis cinerea, the causal agent for gray mold. J Chem Ecol 23:1689–1702. doi:10.1023/B:JOEC.0000006444.79951.75
Bellin D, Peressotti E, Merdinoglu D et al (2009) Resistance to Plasmopara viticola in grapevine “Bianca” is controlled by a major dominant gene causing localised necrosis at the infection site. Theor Appl Genet 120:163–176. doi:10.1007/s00122-009-1167-2
Benikhlef L, L’Haridon F, Abou-Mansour E et al (2013) Perception of soft mechanical stress in Arabidopsis leaves activates disease resistance. BMC Plant Biol 13:133. doi:10.1186/1471-2229-13-133
Boso S, Martínez MC, Unger S, Kassemeyer H-H (2006) Evaluation of foliar resistance to downy mildew in different cv. Albariño clones Vitis - J Grapevine Res 45:23–27
Bouvier F, Backhaus RA, Camara B (1998) Induction and control of chromoplast-specific carotenoid genes by oxidative stress. J Biol Chem 273:30651–30659
Brilli F, Ruuskanen TM, Schnitzhofer R et al (2011) Detection of plant volatiles after leaf wounding and darkening by proton transfer reaction “time-of-flight” mass spectrometry (PTR-TOF). PLoS One 6:e20419. doi:10.1371/journal.pone.0020419
Chiron H, Drouet A, Lieutier F et al (2000) Gene induction of stilbene biosynthesis in scots pine in response to ozone treatment, wounding, and fungal infection. Plant Physiol 124:865–872. doi:10.1104/pp.124.2.865
Chong J, Poutaraud A, Hugueney P (2009) Metabolism and roles of stilbenes in plants. Plant Sci 177:143–155. doi:10.1016/j.plantsci.2009.05.012
Chung I-M, Park MR, Chun JC, Yun SJ (2003) Resveratrol accumulation and resveratrol synthase gene expression in response to abiotic stresses and hormones in peanut plants. Plant Sci 164:103–109. doi:10.1016/S0168-9452(02)00341-2
Conconi A, Miquel M, Browse JA, Ryan CA (1996) Intracellular levels of free linolenic and linoleic acids increase in tomato leaves in response to wounding. Plant Physiol 111:797–803. doi:10.1104/pp.111.3.797
Creelman RA, Mullet JE (1997) Biosynthesis and action of Jasmonates in plants. Annu Rev Plant Physiol Plant Mol Biol 48:355–381. doi:10.1146/annurev.arplant.48.1.355
Csizmazia J, Bereznai L (1968) A szõlõ Plasmopara viticola és a Viteus vitifolii elleni rezisztencia nemesités eredményei. Dans Orszõl Bor Kut Int Evönyve:191–200
Dai GH, Andary C, Cosson-Mondolot L, Boubals D (1994) Polyphenols and resistance of grapevines to downy MILDEW. Acta Hortic 763–766. doi: 10.17660/ActaHortic.1994.381.110
Dai GH, Andary C, Mondolot-Cosson L, Boubals D (1995) Histochemical studies on the interaction between three species of grapevine, Vitis vinifera, V. rupestris and V. rotundifolia and the downy mildew fungus, Plasmopara viticola. Physiol Mol Plant Pathol 46:177–188. doi:10.1006/pmpp.1995.1014
Della Corte A, Chitarrini G, Di Gangi IM et al (2015) A rapid LC-MS/MS method for quantitative profiling of fatty acids, sterols, glycerolipids, glycerophospholipids and sphingolipids in grapes. Talanta 140:52–61. doi:10.1016/j.talanta.2015.03.003
Delye C, Corio-Costet M-F (1998) Origin of primary infections of grape by Uncinula necator: RAPD analysis discriminates two biotypes. Mycol Res 102:283–288. doi:10.1017/S0953756297004632
Dhondt S, Geoffroy P, Stelmach BA et al (2000) Soluble phospholipase A2 activity is induced before oxylipin accumulation in tobacco mosaic virus-infected tobacco leaves and is contributed by patatin-like enzymes. Plant J 23:431–440. doi:10.1046/j.1365-313x.2000.00802.x
Farmer EE, Ryan CA (1992) Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell 4:129–134. doi:10.1105/tpc.4.2.129
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Höll J, Vannozzi A, Czemmel S et al (2013) The R2R3-MYB transcription factors MYB14 and MYB15 regulate stilbene biosynthesis in Vitis vinifera. Plant Cell 25:4135–4149. doi:10.1105/tpc.113.117127
Howe GA, Schilmiller AL (2002) Oxylipin metabolism in response to stress. Curr Opin Plant Biol 5:230–236. doi:10.1016/S1369-5266(02)00250-9
Jailloux F, Thind T, Clerjeau M (1998) Release, germination, and pathogenicity of ascospores of Uncinula necator under controlled conditions. Can J Bot 76:777–781. doi:10.1139/b98-054
Jeandet P, Douillet-Breuil A-C, Bessis R et al (2002) Phytoalexins from the Vitaceae: biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity, and metabolism. J Agric Food Chem 50:2731–2741
Kachroo A, Kachroo P (2009) Fatty acid-derived signals in plant defense. Annu Rev Phytopathol 47:153–176. doi:10.1146/annurev-phyto-080508-081820
Kennelly MM, Gadoury DM, Wilcox WF et al (2007) Primary infection, lesion productivity, and survival of sporangia in the grapevine downy mildew pathogen Plasmopara viticola. Phytopathology 97:512–522. doi:10.1094/PHYTO-97-4-0512
Keshavarz T, Taheri P (2015) Investigating binucleate Rhizoctonia induced defence responses in kidney bean against Rhizoctonia solani. Biocontrol Sci Tech 25:444–459. doi:10.1080/09583157.2014.984285
Kono A, Sato A, Reisch B, Cadle-Davidson L (2015) Effect of detergent on the quantification of grapevine downy mildew sporangia from leaf discs. Hortscience 50:656–660
Kozma P, Dula T (2003) Inheritance of resistance to downy mildew and powdery mildew of hybrid family Muscadinia x V. vinifera x V. amurensis x Franco-American hybrid. Acta Hortic 457–463. doi: 10.17660/ActaHortic.2003.603.58
Langcake P, Pryce RJ (1977) A new class of phytoalexins from grapevines. Experientia 33:151–152. doi:10.1007/BF02124034
Lava SS, Zipper R, Spring O (2015) Sunflower white blister rust—host specificity and fungicide effects on infectivity and early infection stages. Crop Prot 67:214–222. doi:10.1016/j.cropro.2014.10.019
Lebeda A, Mieslerová B, Petrželová I et al (2012) Patterns of virulence variation in the interaction between Lactuca spp. and lettuce powdery mildew (Golovinomyces cichoracearum). Fungal Ecol 5:670–682. doi:10.1016/j.funeco.2012.03.005
Lee S, Suh S, Kim S et al (1997) Systemic elevation of phosphatidic acid and lysophospholipid levels in wounded plants. Plant J 12:547–556. doi:10.1046/j.1365-313X.1997.00547.x
León J, Rojo E, Sánchez-Serrano JJ (2001) Wound signalling in plants. J Exp Bot 52:1–9. doi:10.1093/jexbot/52.354.1
Mattivi F, Vrhovsek U, Malacarne G et al (2011) Profiling of resveratrol oligomers, important stress metabolites, accumulating in the leaves of hybrid Vitis vinifera (Merzling × Teroldego) genotypes infected with Plasmopara viticola. J Agric Food Chem 59:5364–5375. doi:10.1021/jf200771y
McConn M, Creelman RA, Bell E et al (1997) Jasmonate is essential for insect defense in Arabidopsis. Proc Natl Acad Sci 94:5473–5477
Narváez-Vásquez J, Florin-Christensen J, Ryan CA (1999) Positional specificity of a phospholipase a activity induced by wounding, systemin, and oligosaccharide elicitors in tomato leaves. Plant Cell 11:2249–2260. doi:10.1105/tpc.11.11.2249
Noel JP, Austin MB, Bomati EK (2005) Structure–function relationships in plant phenylpropanoid biosynthesis. Curr Opin Plant Biol 8:249–253. doi:10.1016/j.pbi.2005.03.013
Nyadanu D, Akromah R, Adomako B et al (2012) Inheritance and general combining ability studies of detached pod, leaf disc and natural field resistance to Phytophthora palmivora and Phytophthora megakarya in cacao (Theobroma cacao L.) Euphytica 188:253–264. doi:10.1007/s10681-012-0717-x
Park J-W, Balaraju K, Kim J-W et al (2013) Systemic resistance and growth promotion of chili pepper induced by an antibiotic producing Bacillus vallismortis strain BS07. Biol Control 65:246–257. doi:10.1016/j.biocontrol.2013.02.002
Peressotti E, Wiedemann-Merdinoglu S, Delmotte F et al (2010) Breakdown of resistance to grapevine downy mildew upon limited deployment of a resistant variety. BMC Plant Biol 10:147. doi:10.1186/1471-2229-10-147
Pezet R, Gindro K, Viret O, Spring J-L (2004) Glycosylation and oxidative dimerization of resveratrol are respectively associated to sensitivity and resistance of grapevine cultivars to downy mildew. Physiol Mol Plant Pathol 65:297–303. doi:10.1016/j.pmpp.2005.03.002
Prost I, Dhondt S, Rothe G et al (2005) Evaluation of the antimicrobial activities of plant oxylipins supports their involvement in defense against pathogens. Plant Physiol 139:1902–1913. doi:10.1104/pp.105.066274
Reymond P, Weber H, Damond M, Farmer EE (2000) Differential gene expression in response to mechanical wounding and insect feeding in Arabidopsis. Plant Cell 12:707–719. doi:10.1105/tpc.12.5.707
Ryu SB, Wang X (1998) Increase in free linolenic and linoleic acids associated with phospholipase D-mediated hydrolysis of phospholipids in wounded castor bean leaves. Biochim Biophys Acta BBA - Lipids Lipid Metab 1393:193–202. doi:10.1016/S0005-2760(98)00048-4
Savatin DV, Gramegna G, Modesti V, Cervone F (2014) Wounding in the plant tissue: the defense of a dangerous passage. Front Plant Sci. doi:10.3389/fpls.2014.00470
Schnee S, Queiroz EF, Voinesco F et al (2013) Vitis vinifera canes, a new source of antifungal compounds against Plasmopara viticola, Erysiphe necator, and Botrytis cinerea. J Agric Food Chem 61:5459–5467. doi:10.1021/jf4010252
Sun Z-B, Yuan X-F, Zhang H et al (2013) Isolation, screening and identification of antagonistic downy mildew endophytic bacteria from cucumber. Eur J Plant Pathol 137:847–857. doi:10.1007/s10658-013-0293-5
Suthaparan A, Solhaug KA, Stensvand A, Gislerød HR (2016) Determination of UV action spectra affecting the infection process of Oidium neolycopersici, the cause of tomato powdery mildew. J Photochem Photobiol B 156:41–49. doi:10.1016/j.jphotobiol.2016.01.009
Tôyama S (1972) Electron microscope studies on the morphogenesis of plastids. Bot Mag Shokubutsu-Gaku-Zasshi 85:1–10. doi:10.1007/BF02489195
Vannozzi A, Dry IB, Fasoli M et al (2012) Genome-wide analysis of the grapevine stilbene synthase multigenic family: genomic organization and expression profiles upon biotic and abiotic stresses. BMC Plant Biol. doi:10.1186/1471-2229-12-130
Vrhovsek U, Malacarne G, Masuero D et al (2012a) Profiling and accurate quantification of trans-resveratrol, trans-piceid, trans-pterostilbene and 11 viniferins induced by Plasmopara viticola in partially resistant grapevine leaves. Aust J Grape Wine Res 18:11–19. doi:10.1111/j.1755-0238.2011.00163.x
Vrhovsek U, Masuero D, Gasperotti M et al (2012b) A versatile targeted metabolomics method for the rapid quantification of multiple classes of phenolics in fruits and beverages. J Agric Food Chem 60:8831–8840. doi:10.1021/jf2051569
Vu HS, Roston R, Shiva S et al (2015) Modifications of membrane lipids in response to wounding of Arabidopsis thaliana leaves. Plant Signal Behav 10:e1056422. doi:10.1080/15592324.2015.1056422
Wehrens R, Carvalho E, Masuero D et al (2013) High-throughput carotenoid profiling using multivariate curve resolution. Anal Bioanal Chem 405:5075–5086. doi:10.1007/s00216-012-6555-9
Willocquet L, Colombet D, Rougier M et al (1996) Effects of radiation, especially ultraviolet B, on conidial germination and mycelial growth of grape powdery mildew. Eur J Plant Pathol 102:441–449. doi:10.1007/BF01877138
Zhang S, Yu S, Zhang F et al (2012) Inheritance of downy mildew resistance at different developmental stages in Chinese cabbage via the leaf disk test. Hortic Environ Biotechnol 53:397–403. doi:10.1007/s13580-012-0057-8
Zhao X, Ren L, Yin H et al (2013) Sensitivity of Pseudoperonospora cubensis to dimethomorph, metalaxyl and fosetyl-aluminium in Shanxi of China. Crop Prot 43:38–44. doi:10.1016/j.cropro.2012.08.002
Acknowledgements
This research was supported by the FIRST International PhD program and ADP 2015/2016 project founded by the Autonomous Province of Trento.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Hanns H. Kassemeyer
Electronic supplementary material
ESM 1
(XLSX 39.2 kb)
Rights and permissions
About this article
Cite this article
Chitarrini, G., Zulini, L., Masuero, D. et al. Lipid, phenol and carotenoid changes in ‘Bianca’ grapevine leaves after mechanical wounding: a case study. Protoplasma 254, 2095–2106 (2017). https://doi.org/10.1007/s00709-017-1100-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-017-1100-5